UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the
Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): October 2, 2025
(Exact Name of Registrant as Specified in Charter)
|
|
|
|
|
(State or Other Jurisdiction of Incorporation)
|
(Commission File Number)
|
(IRS Employer Identification No.)
|
|
|
|
|
|
(Address of Principal Executive Offices)
|
(Zip Code)
|
(518 ) 795-1400
(Registrant’s telephone number, including area code)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under
any of the following provisions:
|
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
|
|
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
|
|
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2 (b))
|
|
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4 (c))
|
Securities registered pursuant to Section 12(b) of the Act:
|
Title of each class
|
Trading Symbol(s)
|
Name of each exchange on which
registered
|
|
|
|
|
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2
of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised
financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 2.02 – Results of Operations and Financial Condition.
On October 2, 2025, AngioDynamics, Inc. (“AngioDynamics”) issued a press release announcing financial results for the fiscal first quarter ended
August 31, 2025. A copy of the press release is furnished herewith as Exhibit 99.1.
The information set forth in Item 2.02 of this Form 8-K (including Exhibit 99.1) shall not be deemed “filed” for purposes of Section 18 of the
Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities under that Section. Furthermore, such information shall not be deemed to be incorporated by reference into any filing under the Securities Act
of 1933, as amended (the “Securities Act”), or the Exchange Act, except as shall be expressly set forth by specific reference in such a filing.
Item 7.01 – Regulation FD Disclosure.
Presentation slides discussing AngioDynamics and its fiscal first quarter ended August 31, 2025 are furnished herewith as Exhibit 99.2.
The presentation slides furnished pursuant to Item 7.01 of this Form 8-K (including Exhibit 99.2) shall not be deemed “filed” for purposes of
Section 18 of the Exchange Act, or otherwise subject to the liabilities under that Section. Furthermore, the presentation slides shall not be deemed to be incorporated by reference into any filing under the Securities Act or the Exchange Act.
Forward-Looking Statements
This document and its attachments contain forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All
statements regarding AngioDynamics’ expected future financial position, results of operations, cash flows, business strategy, budgets, projected costs, capital expenditures, products, competitive positions, growth opportunities, plans and
objectives of management for future operations, as well as statements that include the words such as “expects,” “reaffirms,” “intends,” “anticipates,” “plans,” “believes,” “seeks,” “estimates,” “projects”, “optimistic,” or variations of such words
and similar expressions, are forward-looking statements. These forward-looking statements are not guarantees of future performance and are subject to risks and uncertainties. Investors are cautioned that actual events or results may differ
materially from AngioDynamics’ expectations, expressed or implied. Factors that may affect the actual results achieved by AngioDynamics include, without limitation, the scale and scope of the COVID-19 global pandemic, the ability of AngioDynamics
to develop its existing and new products, technological advances and patents attained by competitors, infringement of AngioDynamics’ technology or assertions that AngioDynamics’ technology infringes the technology of third parties, the ability of
AngioDynamics to effectively compete against competitors that have substantially greater resources, future actions by the FDA or other regulatory agencies, domestic and foreign health care reforms and government regulations, results of pending or
future clinical trials, overall economic conditions (including inflation, tariffs, labor shortages and supply chain challenges including the cost and availability of raw materials), the results of on-going litigation, challenges with respect to
third-party distributors or joint venture partners or collaborators, the results of sales efforts, the effects of product recalls and product liability claims, changes in key personnel, the ability of AngioDynamics to execute on strategic
initiatives, the effects of economic, credit and capital market conditions, general market conditions, market acceptance, foreign currency exchange rate fluctuations, the effects on pricing from group purchasing organizations and competition, the
ability of AngioDynamics to obtain regulatory clearances or approval of its products, or to integrate acquired businesses, as well as the risk factors listed from time to time in AngioDynamics’ SEC filings, including but not limited to its Annual
Report on Form 10-K for the year ended May 31, 2025. AngioDynamics does not assume any obligation to publicly update or revise any forward-looking statements for any reason.
Item 9.01 – Financial Statements and Exhibits.
| (d) |
Exhibits.
|
|
Exhibit No.
|
Description
|
|
Press Release, dated October 2, 2025.
|
|
|
Presentation, dated October 2, 2025.
|
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on
its behalf by the undersigned hereunto duly authorized.
|
ANGIODYNAMICS, INC.
|
||
|
(Registrant)
|
||
|
Date: October 2, 2025
|
By: /s/ Lawrence T. Weiss
|
|
|
Name:
|
Lawrence T. Weiss
|
|
|
Title:
|
Senior Vice President, Chief Legal Officer and Corporate Secretary
|
|
Exhibit 99.1
AngioDynamics Reports Fiscal Year 2026 First Quarter Financial Results; Med Tech Growth of 26.1% Drives Continued Momentum
| • |
Med Tech segment delivers fourth consecutive quarter of over 20% revenue growth
|
| • |
Reported Adjusted EBITDA of $2.2 million compared to ($0.2) million in the prior year
|
| • |
Continues to expect to be cash flow positive for the full fiscal year 2026
|
| • |
Raised full year FY 2026 guidance for net sales, Med Tech net sales growth, Adjusted EBITDA and Adjusted EPS
|
LATHAM, N.Y.--(BUSINESS WIRE)— Oct. 2, 2025-- AngioDynamics, Inc. (NASDAQ: ANGO), a leading and transformative medical technology company focused on restoring healthy
blood flow in the body’s vascular system, expanding cancer treatment options, and improving quality of life for patients, today announced financial results for the first quarter of fiscal year 2026, which ended August 31, 2025.
Fiscal Year 2026 First Quarter Highlights
|
Quarter Ended
August 31, 2025
|
Pro Forma* YoY Growth
|
|
|
Net Sales
|
$75.7 million
|
12.2%
|
|
Med Tech Net Sales
|
$35.3 million
|
26.1%
|
|
Med Device Net Sales
|
$40.4 million
|
2.3%
|
| • |
GAAP gross margin of 55.3%
|
| • |
GAAP loss per share of $0.26
|
| • |
Adjusted loss per share of $0.10
|
| • |
Adjusted EBITDA of $2.2 million
|
| • |
Ended fiscal 2026 first quarter with $38.8 million in cash and cash equivalents, ahead of expectations, continues to expect to be cash flow positive for the full year FY 2026
|
| • |
First patients enrolled in both the AMBITION BTK and RECOVER-AV trials
|
| • |
NanoKnife PRESERVE study published in the journal of European Urology
|
*Pro forma results exclude the Dialysis and BioSentry businesses divested in June 2023 and the PICC, Midline and tip location product portfolios
divested in February 2024, as well as the discontinued Radiofrequency and Syntrax support catheter products in February 2024.
"We had an outstanding first quarter as we continued to build off of the strong momentum created in fiscal 2025," commented Jim Clemmer,
President and Chief Executive Officer of AngioDynamics, Inc. "Our strategy to bring unique platform technologies to large, fast growing global markets has paid off as we reported our fourth quarter in a row of MedTech growth of over 20%. This
continued performance, combined with our disciplined focus on operational excellence, is driving sustained profitable growth.”
1
Mr. Clemmer continued, “Our exceptional team is executing our vision to deliver transformative technologies that expand treatment options and
help patients live healthier, happier lives. With our superior technologies supported by our clinical investments and the strength of our balance sheet, we remain well-positioned to drive consistent, profitable growth, and deliver sustained value
creation during the balance of 2026 and beyond.”
Fiscal Year 2026 First Quarter Financial Results
Unless otherwise noted, all financial comparisons below are presented on a pro forma basis excluding the Dialysis and BioSentry businesses
divested in June 2023, the PICC, Midline, and tip location product portfolios divested in February 2024, and the RadioFrequency and Syntrax support catheter products discontinued in February 2024.
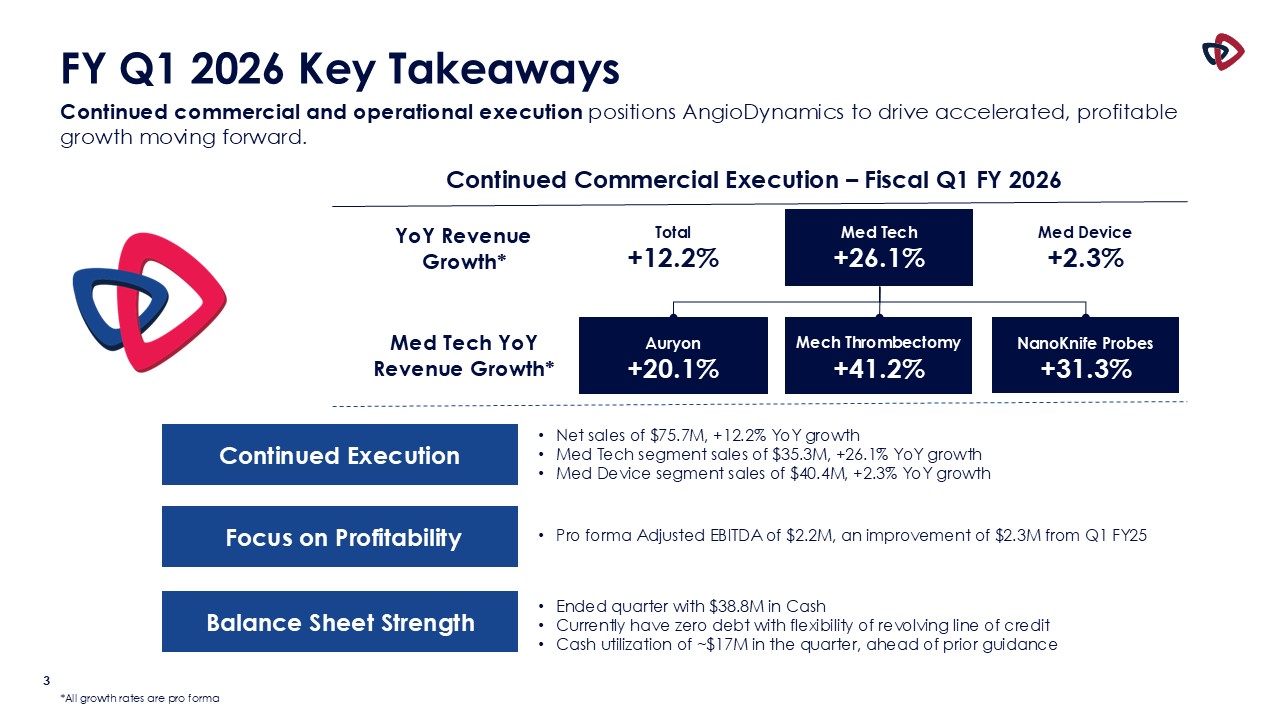
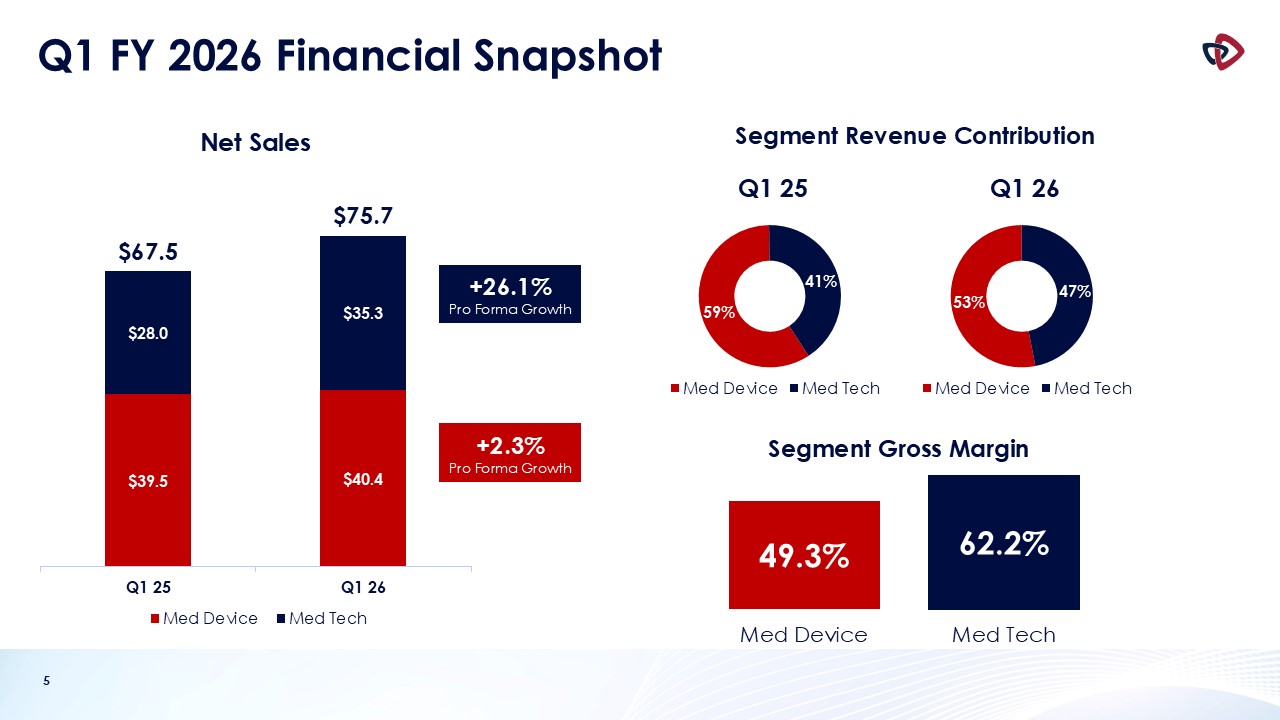
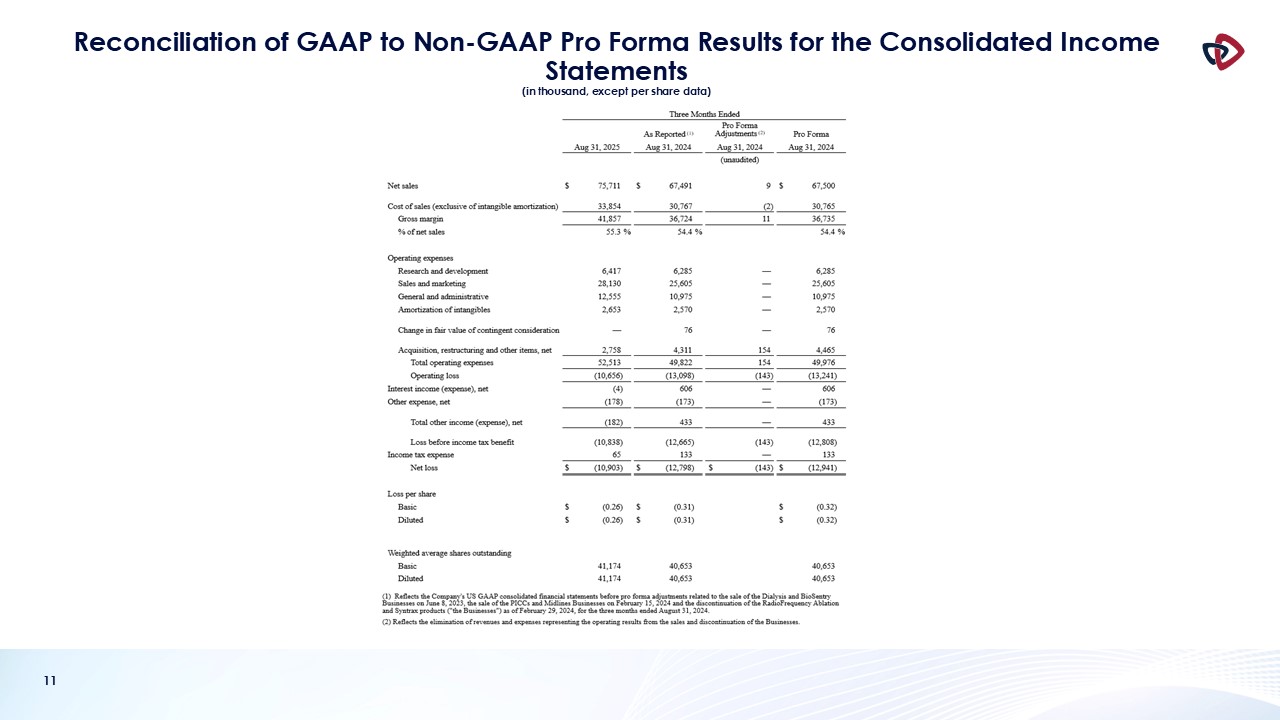
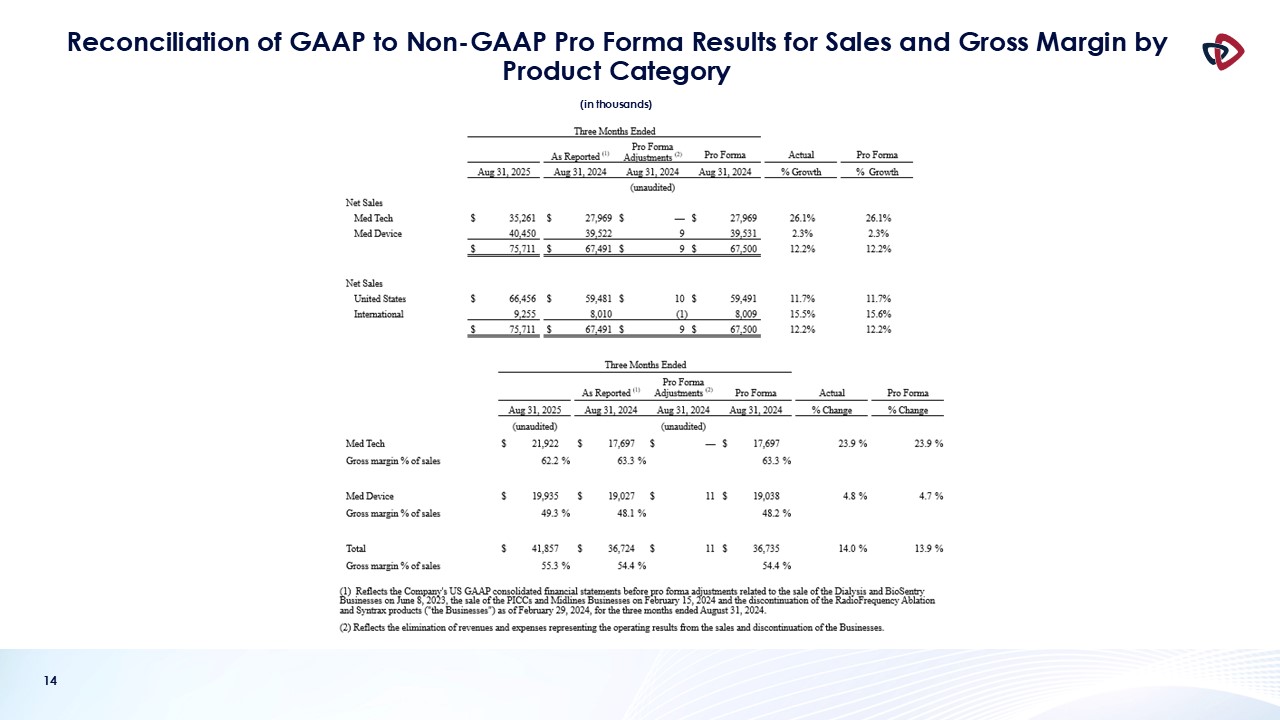
Net sales for the first quarter of fiscal year 2026 were $75.7 million, an increase of 12.2% compared to the prior-year quarter.
Med Tech net sales were $35.3 million, a 26.1% increase from $28.0 million in the prior-year period. Med Tech includes the Auryon peripheral
atherectomy platform, the thrombus management platform and the NanoKnife irreversible electroporation platform.
Growth during the quarter was driven by solid performance across the Med Tech segment. Auryon sales were $16.5 million an increase of 20.1%, our
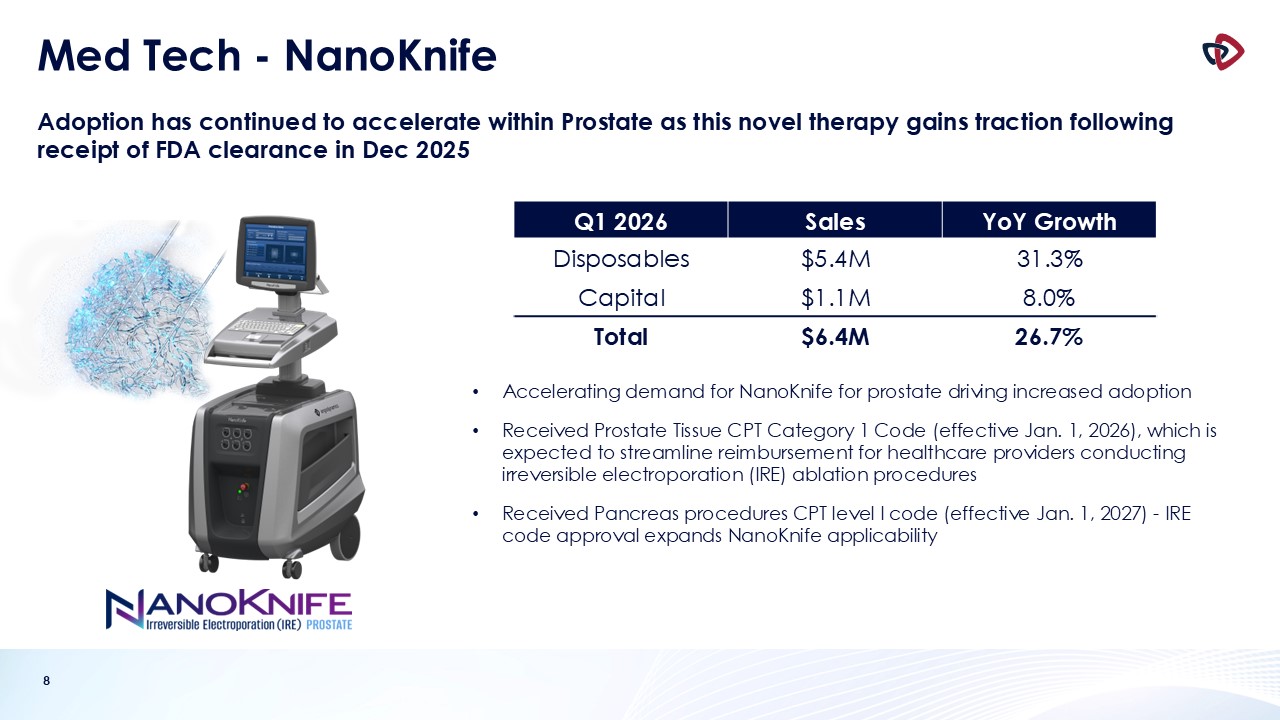
Mechanical Thrombectomy business, which includes AngioVac and AlphaVac, delivered sales of $11.3 million, an increase of 41.2%, and NanoKnife sales were $6.4 million, an increase of 26.7%, including 31.3% growth in probes.
Med Device net sales were $40.4 million, a 2.3% increase compared to $39.5 million in the prior-year period.
Gross margin for the first quarter of fiscal 2026 was 55.3%, which was 90 basis points higher compared to the first quarter of fiscal 2025, and
260 basis points higher sequentially from 52.7% in the fourth quarter of fiscal 2025, primarily due to increased Med Tech revenue, as well as operational efficiencies. Gross margin included $1.7 million of tariff expense.
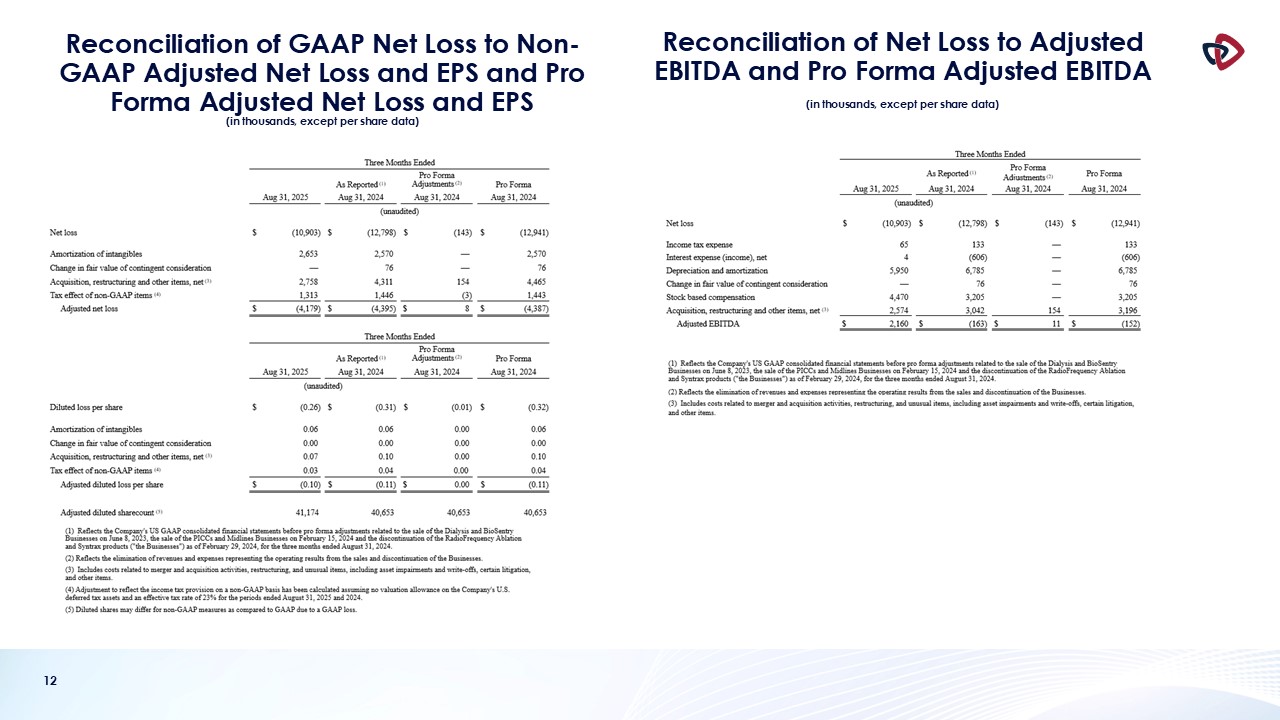
The Company recorded a GAAP net loss of $10.9 million, or a loss per share of $0.26, in the first quarter of fiscal 2026. Excluding the items
shown in the non-GAAP reconciliation table below, adjusted net loss for the first quarter of fiscal 2026 was $4.2 million, or a loss per share of $0.10. This compares to an adjusted net loss during the fiscal first quarter of 2025 of $4.4
million, or a loss per share of $0.11.
2
Adjusted EBITDA in the first quarter of fiscal 2026, excluding the items shown in the non-GAAP reconciliation table below, was $2.2 million,
compared to $(0.2) million in the first quarter of fiscal 2025.
In the first quarter of fiscal 2026, the Company used $17.1 million of cash. The Company’s first fiscal quarter has historically exhibited the
highest utilization of cash during the year, and the first quarter of fiscal 2026 was better than the Company’s expectations, resulting in the use of less cash than expected. The Company continues to expect to be cash flow positive for the full
year fiscal 2026.
At August 31, 2025, the Company had $38.8 million in cash and cash equivalents compared to $55.9 million in cash and cash equivalents at May 31,
2025. The Company maintains a debt-free balance sheet.
First Patient Enrolled in AMBITION BTK Trial
During the quarter, the Company announced that it achieved a significant clinical milestone with the enrollment of the first patient in the
AMBITION BTK trial, a prospective, multicenter, randomized controlled trial designed to investigate the clinical safety and effectiveness of the Auryon Atherectomy System in treating challenging below-the-knee lesions in patients with Critical
Limb Ischemia. The trial will include up to 224 patients at up to 30 sites, with a companion registry enrolling up to 1,500 additional patients, representing the Company's continued commitment to advancing clinical evidence for the treatment of
peripheral artery disease and expanding the clinical applications for the Auryon platform.
First Patient Enrolled in RECOVER-AV Clinical Trial
During the quarter, the Company announced the first patient enrollment in the RECOVER-AV clinical trial, a prospective, multi-center,
multi-national, single-arm study evaluating the AlphaVac F1885 System for the treatment of acute, intermediate-risk pulmonary embolism. The study builds on the existing U.S. FDA 510(k) clearance and European CE Mark approval to assess mechanical
thrombectomy treatment and long-term functional outcomes in intermediate-risk PE patients across Europe, Canada, and Hong Kong. The trial will enroll patients at up to 20 hospital-based sites and follows patients for 12 months, with functional
and quality-of-life outcomes assessed at 30 days and 12 months, representing the Company's continued commitment to expanding its global clinical presence and generating evidence-based data to support broader access to life-saving PE treatment.
3
NanoKnife PRESERVE Study Results Published in Leading European Journal
During the quarter, the Company announced the publication of results from the PRESERVE study in European Urology, a leading journal in urologic
research. The study assessed the safety and effectiveness of irreversible electroporation with the NanoKnife System to ablate prostate tissue in patients with intermediate-risk prostate cancer. The PRESERVE clinical study met its primary
effectiveness endpoint, with 84.0% of men free from in-field, clinically significant disease at 12 months post-procedure. The study also demonstrated strong quality-of-life outcomes, with urinary continence largely preserved (97% at baseline vs.
96% at 12 months) and 84% of patients with good baseline sexual function at 12 months, reinforcing the NanoKnife System's role in providing effective treatment while preserving quality of life.
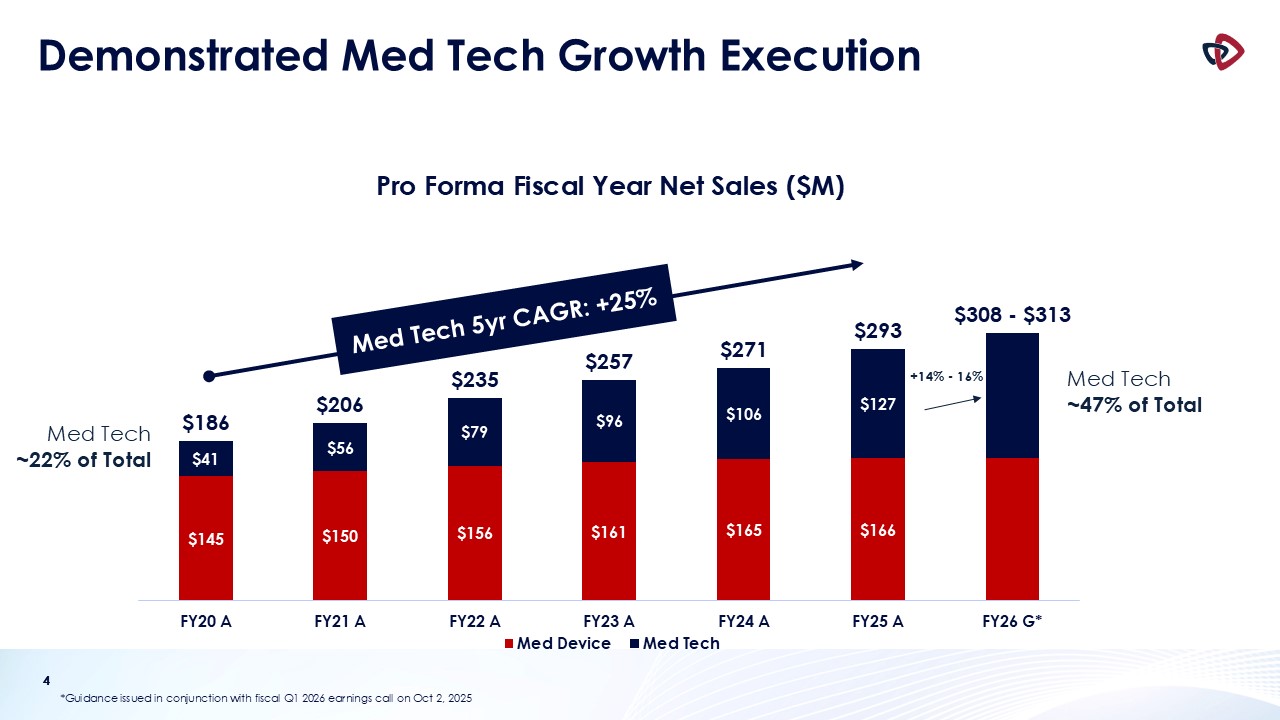
Fiscal Year 2026 Financial Guidance
For fiscal year 2026 the company now expects:
|
Guidance Metric
|
Guidance Action
|
Current Guidance
(as of October 2, 2025)
|
Previous Guidance
(as of July 15, 2025)
|
|
Net Sales
|
Increased
|
$308 - $313 million
|
$305 - $310 million
|
|
Med Tech Net Sales Growth
|
Increased
|
14% - 16%
|
12% - 15%
|
|
Med Device Net Sales Growth
|
Unchanged
|
Flat
|
Flat
|
|
Gross Margin
|
Unchanged
|
53.5% - 55.5%
|
53.5% - 55.5%
|
|
Adjusted EBITDA
|
Increased
|
$6.0 - $10.0 million
|
$3.0 - $8.0 million
|
|
Adjusted EPS
|
Increased
|
($0.33) – ($0.23)
|
($0.35) – ($0.25)
|
|
Free Cash Flow
|
Unchanged
|
Positive for full year FY 2026
|
Positive for full year FY 2026
|
Tariff Related Guidance Assumptions
For the full fiscal year 2026, the company continues to expect a $4.0 - $6.0 million impact from tariffs, which are included in the above
provided guidance.
All assumptions made related to expected tariff impacts are based on the Company’s point of view on the current tariff situation, as of October
2, 2025. As the situation is fluid, these assumptions may change in the future.
Conference Call
The Company’s management will host a conference call at 8:00 a.m. ET the same day to discuss the results. To participate in the conference call,
dial 1-877-407-0784 (domestic) or +1-201-689-8560 (international). This conference call will also be webcast and can be accessed from the “Investors” section of the AngioDynamics website at www.angiodynamics.com. The webcast replay of the call
will be available at the same site approximately one hour after the end of the call.
A recording of the call will also be available, until Thursday, October 09, 2025 at 11:59 PM ET. To hear this recording, dial 1-844-512-2921
(domestic) or +1-412-317-6671 (international) and enter the passcode 13755707.
4
Use of Non-GAAP Measures
Management uses non-GAAP measures to establish operational goals and believes that non-GAAP measures may assist investors in analyzing the
underlying trends in AngioDynamics' business over time. Investors should consider these non-GAAP measures in addition to, not as a substitute for or as superior to, financial reporting measures prepared in accordance with GAAP. In this news
release, AngioDynamics has reported pro forma results, adjusted EBITDA, adjusted net income and adjusted earnings per share. Management uses these measures in its internal analysis and review of operational performance. Management believes that
these measures provide investors with useful information in comparing AngioDynamics' performance over different periods. By using these non-GAAP measures, management believes that investors get a better picture of the performance of
AngioDynamics' underlying business. Management encourages investors to review AngioDynamics' financial results prepared in accordance with GAAP to understand AngioDynamics' performance taking into account all relevant factors, including those
that may only occur from time to time but have a material impact on AngioDynamics' financial results. Please see the tables that follow for a reconciliation of non-GAAP measures to measures prepared in accordance with GAAP.
About AngioDynamics, Inc.
AngioDynamics is a leading and transformative medical technology company focused on restoring healthy blood flow in the body’s vascular system,
expanding cancer treatment options and improving quality of life for patients.
The Company’s innovative technologies and devices are chosen by talented
physicians in fast-growing healthcare markets to treat unmet patient needs. For more information, visit www.angiodynamics.com.
5
Safe Harbor
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements
regarding AngioDynamics' expected future financial position, results of operations, cash flows, business strategy, budgets, projected costs, capital expenditures, products, competitive positions, growth opportunities, plans and objectives of
management for future operations, as well as statements that include the words such as "expects," "reaffirms," "intends," "anticipates," "plans," "believes," "seeks," "estimates," "projects," "optimistic," or variations of such words and similar
expressions, are forward-looking statements. These forward-looking statements are not guarantees of future performance and are subject to risks and uncertainties. Investors are cautioned that actual events or results may differ materially from
AngioDynamics' expectations, expressed or implied. Factors that may affect the actual results achieved by AngioDynamics include, without limitation, the scale and scope of the COVID-19 global pandemic, the ability of AngioDynamics to develop its
existing and new products, technological advances and patents attained by competitors, infringement of AngioDynamics' technology or assertions that AngioDynamics' technology infringes the technology of third parties, the ability of AngioDynamics
to effectively compete against competitors that have substantially greater resources, future actions by the FDA or other regulatory agencies, domestic and foreign health care reforms and government regulations, results of pending or future
clinical trials, overall economic conditions (including inflation, tariffs, labor shortages and supply chain challenges including the cost and availability of raw materials), the results of on-going litigation, challenges with respect to
third-party distributors or joint venture partners or collaborators, the results of sales efforts, the effects of product recalls and product liability claims, changes in key personnel, the ability of AngioDynamics to execute on strategic
initiatives, the effects of economic, credit and capital market conditions, general market conditions, market acceptance, foreign currency exchange rate fluctuations, the effects on pricing from group purchasing organizations and competition, the
ability of AngioDynamics to obtain regulatory clearances or approval of its products, or to integrate acquired businesses, as well as the risk factors listed from time to time in AngioDynamics' SEC filings, including but not limited to its Annual
Report on Form 10-K for the year ended May 31, 2025. AngioDynamics does not assume any obligation to publicly update or revise any forward-looking statements for any reason.
Investors:
Stephen Trowbridge
Executive Vice President & CFO
518-795-1408
strowbridge@angiodynamics.com
Media:
Saleem Cheeks
Vice President, Communications
518-795-1174
scheeks@angiodynamics.com
6
ANGIODYNAMICS, INC. AND SUBSIDIARIES
CONSOLIDATED INCOME STATEMENTS
(in thousands, except per share data)
|
Three Months Ended
|
||||||||||||||||
|
As Reported (1)
|
Pro Forma
Adjustments (2)
|
Pro Forma
|
||||||||||||||
|
Aug 31, 2025
|
Aug 31, 2024
|
Aug 31, 2024
|
Aug 31, 2024
|
|||||||||||||
|
(unaudited)
|
||||||||||||||||
|
Net sales
|
$
|
75,711
|
$
|
67,491
|
9
|
$
|
67,500
|
|||||||||
|
Cost of sales (exclusive of intangible amortization)
|
33,854
|
30,767
|
(2
|
)
|
30,765
|
|||||||||||
|
Gross margin
|
41,857
|
36,724
|
11
|
36,735
|
||||||||||||
|
% of net sales
|
55.3
|
%
|
54.4
|
%
|
54.4
|
%
|
||||||||||
|
Operating expenses
|
||||||||||||||||
|
Research and development
|
6,417
|
6,285
|
—
|
6,285
|
||||||||||||
|
Sales and marketing
|
28,130
|
25,605
|
—
|
25,605
|
||||||||||||
|
General and administrative
|
12,555
|
10,975
|
—
|
10,975
|
||||||||||||
|
Amortization of intangibles
|
2,653
|
2,570
|
—
|
2,570
|
||||||||||||
|
Change in fair value of contingent consideration
|
—
|
76
|
—
|
76
|
||||||||||||
|
Acquisition, restructuring and other items, net
|
2,758
|
4,311
|
154
|
4,465
|
||||||||||||
|
Total operating expenses
|
52,513
|
49,822
|
154
|
49,976
|
||||||||||||
|
Operating loss
|
(10,656
|
)
|
(13,098
|
)
|
(143
|
)
|
(13,241
|
)
|
||||||||
|
Interest income (expense), net
|
(4
|
)
|
606
|
—
|
606
|
|||||||||||
|
Other expense, net
|
(178
|
)
|
(173
|
)
|
—
|
(173
|
)
|
|||||||||
|
Total other income (expense), net
|
(182
|
)
|
433
|
—
|
433
|
|||||||||||
|
Loss before income tax benefit
|
(10,838
|
)
|
(12,665
|
)
|
(143
|
)
|
(12,808
|
)
|
||||||||
|
Income tax expense
|
65
|
133
|
—
|
133
|
||||||||||||
|
Net loss
|
$
|
(10,903
|
)
|
$
|
(12,798
|
)
|
$
|
(143
|
)
|
$
|
(12,941
|
)
|
||||
|
Loss per share
|
||||||||||||||||
|
Basic
|
$
|
(0.26
|
)
|
$
|
(0.31
|
)
|
$
|
(0.32
|
)
|
|||||||
|
Diluted
|
$
|
(0.26
|
)
|
$
|
(0.31
|
)
|
$
|
(0.32
|
)
|
|||||||
|
Weighted average shares outstanding
|
||||||||||||||||
|
Basic
|
41,174
|
40,653
|
40,653
|
|||||||||||||
|
Diluted
|
41,174
|
40,653
|
40,653
|
|||||||||||||
(1) Reflects the Company's US GAAP consolidated financial statements before pro forma adjustments related to the sale of the Dialysis and BioSentry Businesses on June 8,
2023, the sale of the PICCs and Midlines Businesses on February 15, 2024 and the discontinuation of the RadioFrequency Ablation and Syntrax products ("the Businesses") as of February 29, 2024, for the three months ended August 31, 2024.
(2) Reflects the elimination of revenues and expenses representing the operating results from the sales and discontinuation of the Businesses.
7
ANGIODYNAMICS, INC. AND SUBSIDIARIES
GAAP TO NON-GAAP RECONCILIATION
(in thousands, except per share data)
Reconciliation of Net Loss to non-GAAP Adjusted Net Loss and Pro Forma Adjusted Net Loss:
|
Three Months Ended
|
||||||||||||||||
|
As Reported (1)
|
Pro Forma
Adjustments (2)
|
Pro Forma
|
||||||||||||||
|
Aug 31, 2025
|
Aug 31, 2024
|
Aug 31, 2024
|
Aug 31, 2024
|
|||||||||||||
|
(unaudited)
|
||||||||||||||||
|
Net loss
|
$
|
(10,903
|
)
|
$
|
(12,798
|
)
|
$
|
(143
|
)
|
$
|
(12,941
|
)
|
||||
|
Amortization of intangibles
|
2,653
|
2,570
|
—
|
2,570
|
||||||||||||
|
Change in fair value of contingent consideration
|
—
|
76
|
—
|
76
|
||||||||||||
|
Acquisition, restructuring and other items, net (3)
|
2,758
|
4,311
|
154
|
4,465
|
||||||||||||
|
Tax effect of non-GAAP items (4)
|
1,313
|
1,446
|
(3
|
)
|
1,443
|
|||||||||||
|
Adjusted net loss
|
$
|
(4,179
|
)
|
$
|
(4,395
|
)
|
$
|
8
|
$
|
(4,387
|
)
|
|||||
Reconciliation of Diluted Loss and Pro Forma Diluted Loss Per Share to non-GAAP Adjusted and Pro Forma Adjusted Diluted Loss Per
Share:
|
Three Months Ended
|
||||||||||||||||
|
As Reported (1)
|
Pro Forma
Adjustments (2)
|
Pro Forma
|
||||||||||||||
|
Aug 31, 2025
|
Aug 31, 2024
|
Aug 31, 2024
|
Aug 31, 2024
|
|||||||||||||
|
(unaudited)
|
||||||||||||||||
|
Diluted loss per share
|
$
|
(0.26
|
)
|
$
|
(0.31
|
)
|
$
|
(0.01
|
)
|
$
|
(0.32
|
)
|
||||
|
Amortization of intangibles
|
0.06
|
0.06
|
0.00
|
0.06
|
||||||||||||
|
Change in fair value of contingent consideration
|
0.00
|
0.00
|
0.00
|
0.00
|
||||||||||||
|
Acquisition, restructuring and other items, net (3)
|
0.07
|
0.10
|
0.00
|
0.10
|
||||||||||||
|
Tax effect of non-GAAP items (4)
|
0.03
|
0.04
|
0.00
|
0.04
|
||||||||||||
|
Adjusted diluted loss per share
|
$
|
(0.10
|
)
|
$
|
(0.11
|
)
|
$
|
0.00
|
$
|
(0.11
|
)
|
|||||
|
Adjusted diluted sharecount (5)
|
41,174
|
40,653
|
40,653
|
40,653
|
||||||||||||
(1) Reflects the Company's US GAAP consolidated financial statements before pro forma adjustments related to the sale of the Dialysis and BioSentry Businesses on June 8,
2023, the sale of the PICCs and Midlines Businesses on February 15, 2024 and the discontinuation of the RadioFrequency Ablation and Syntrax products ("the Businesses") as of February 29, 2024, for the three months ended August 31, 2024.
(2) Reflects the elimination of revenues and expenses representing the operating results from the sales and discontinuation of the Businesses.
(3) Includes costs related to merger and acquisition activities, restructuring, and unusual items, including asset impairments and write-offs,
certain litigation, and other items.
(4) Adjustment to reflect the income tax provision on a non-GAAP basis has been calculated assuming no valuation allowance on the Company's U.S. deferred tax assets and
an effective tax rate of 23% for the periods ended August 31, 2025 and 2024.
(5) Diluted shares may differ for non-GAAP measures as compared to GAAP due to a GAAP loss.
8
ANGIODYNAMICS, INC. AND SUBSIDIARIES
GAAP TO NON-GAAP RECONCILIATION (Continued)
(in thousands, except per share data)
Reconciliation of Net Loss and Pro Forma Net Loss to Adjusted EBITDA and Pro Forma Adjusted EBITDA:
|
Three Months Ended
|
||||||||||||||||
|
As Reported (1)
|
Pro Forma
Adjustments (2)
|
Pro Forma
|
||||||||||||||
|
Aug 31, 2025
|
Aug 31, 2024
|
Aug 31, 2024
|
Aug 31, 2024
|
|||||||||||||
|
(unaudited)
|
||||||||||||||||
|
Net loss
|
$
|
(10,903
|
)
|
$
|
(12,798
|
)
|
$
|
(143
|
)
|
$
|
(12,941
|
)
|
||||
|
Income tax expense
|
65
|
133
|
—
|
133
|
||||||||||||
|
Interest expense (income), net
|
4
|
(606
|
)
|
—
|
(606
|
)
|
||||||||||
|
Depreciation and amortization
|
5,950
|
6,785
|
—
|
6,785
|
||||||||||||
|
Change in fair value of contingent consideration
|
—
|
76
|
—
|
76
|
||||||||||||
|
Stock based compensation
|
4,470
|
3,205
|
—
|
3,205
|
||||||||||||
|
Acquisition, restructuring and other items, net (3)
|
2,574
|
3,042
|
154
|
3,196
|
||||||||||||
|
Adjusted EBITDA
|
$
|
2,160
|
$
|
(163
|
)
|
$
|
11
|
$
|
(152
|
)
|
||||||
(1) Reflects the Company's US GAAP consolidated financial statements before pro forma adjustments related to the sale of the Dialysis and BioSentry Businesses on June 8,
2023, the sale of the PICCs and Midlines Businesses on February 15, 2024 and the discontinuation of the RadioFrequency Ablation and Syntrax products ("the Businesses") as of February 29, 2024, for the three months ended August 31, 2024.
(2) Reflects the elimination of revenues and expenses representing the operating results from the sales and discontinuation of the Businesses.
(3) Includes costs related to merger and acquisition activities, restructuring, and unusual items, including asset impairments and write-offs,
certain litigation, and other items.
9
ANGIODYNAMICS, INC. AND SUBSIDIARIES
ACQUISITION, RESTRUCTURING, AND OTHER ITEMS, NET DETAIL
(in thousands)
|
Three Months Ended
|
||||||||
|
(in thousands)
|
Aug 31, 2025
|
Aug 31, 2024
|
||||||
|
Legal (1)
|
$
|
213
|
$
|
507
|
||||
|
Plant closure (2)
|
2,345
|
3,589
|
||||||
|
Transition service agreement (3)
|
(302
|
)
|
(507
|
)
|
||||
|
Other
|
502
|
722
|
||||||
|
Total
|
$
|
2,758
|
$
|
4,311
|
||||
| (1) |
Legal expenses related to litigation that is outside the normal course of business.
|
| (2) |
Plant closure expense, related to the restructuring of our manufacturing footprint which was announced on January 5, 2024.
|
| (3) |
Transition services agreements that were entered into with Merit and Spectrum.
|
10
ANGIODYNAMICS, INC. AND SUBSIDIARIES
NET SALES BY PRODUCT CATEGORY AND BY GEOGRAPHY
(in thousands)
|
Three Months Ended
|
||||||||||||||||||||||||
|
As Reported (1)
|
Pro Forma
Adjustments (2)
|
Pro Forma
|
Actual
|
Pro Forma
|
||||||||||||||||||||
|
Aug 31, 2025
|
Aug 31, 2024
|
Aug 31, 2024
|
Aug 31, 2024
|
% Growth
|
% Growth
|
|||||||||||||||||||
|
(unaudited)
|
||||||||||||||||||||||||
|
Net Sales
|
||||||||||||||||||||||||
|
Med Tech
|
$
|
35,261
|
$
|
27,969
|
$
|
—
|
$
|
27,969
|
26.1
|
%
|
26.1
|
%
|
||||||||||||
|
Med Device
|
40,450
|
39,522
|
9
|
39,531
|
2.3
|
%
|
2.3
|
%
|
||||||||||||||||
|
$
|
75,711
|
$
|
67,491
|
$
|
9
|
$
|
67,500
|
12.2
|
%
|
12.2
|
%
|
|||||||||||||
|
Net Sales
|
||||||||||||||||||||||||
|
United States
|
$
|
66,456
|
$
|
59,481
|
$
|
10
|
$
|
59,491
|
11.7
|
%
|
11.7
|
%
|
||||||||||||
|
International
|
9,255
|
8,010
|
(1
|
)
|
8,009
|
15.5
|
%
|
15.6
|
%
|
|||||||||||||||
|
$
|
75,711
|
$
|
67,491
|
$
|
9
|
$
|
67,500
|
12.2
|
%
|
12.2
|
%
|
|||||||||||||
(1) Reflects the Company's US GAAP consolidated financial statements before pro forma adjustments related to the sale of the Dialysis and BioSentry Businesses on June 8,
2023, the sale of the PICCs and Midlines Businesses on February 15, 2024 and the discontinuation of the RadioFrequency Ablation and Syntrax products ("the Businesses") as of February 29, 2024, for the three months ended August 31, 2024.
(2) Reflects the elimination of revenues and expenses representing the operating results from the sales and discontinuation of the Businesses.
GROSS MARGIN BY PRODUCT CATEGORY
(in thousands)
|
Three Months Ended
|
||||||||||||||||||||||||
|
As Reported (1)
|
Pro Forma
Adjustments (2)
|
Pro Forma
|
Actual
|
Pro Forma
|
||||||||||||||||||||
|
Aug 31, 2025
|
Aug 31, 2024
|
Aug 31, 2024
|
Aug 31, 2024
|
% Change
|
% Change
|
|||||||||||||||||||
|
(unaudited)
|
(unaudited)
|
|||||||||||||||||||||||
|
Med Tech
|
$
|
21,922
|
$
|
17,697
|
$
|
—
|
$
|
17,697
|
23.9
|
%
|
23.9
|
%
|
||||||||||||
|
Gross margin % of sales
|
62.2
|
%
|
63.3
|
%
|
63.3
|
%
|
||||||||||||||||||
|
Med Device
|
$
|
19,935
|
$
|
19,027
|
$
|
11
|
$
|
19,038
|
4.8
|
%
|
4.7
|
%
|
||||||||||||
|
Gross margin % of sales
|
49.3
|
%
|
48.1
|
%
|
48.2
|
%
|
||||||||||||||||||
|
Total
|
$
|
41,857
|
$
|
36,724
|
$
|
11
|
$
|
36,735
|
14.0
|
%
|
13.9
|
%
|
||||||||||||
|
Gross margin % of sales
|
55.3
|
%
|
54.4
|
%
|
54.4
|
%
|
||||||||||||||||||
(1) Reflects the Company's US GAAP consolidated financial statements before pro forma adjustments related to the sale of the Dialysis and BioSentry Businesses on June 8,
2023, the sale of the PICCs and Midlines Businesses on February 15, 2024 and the discontinuation of the RadioFrequency Ablation and Syntrax products ("the Businesses") as of February 29, 2024, for the three months ended August 31, 2024.
(2) Reflects the elimination of revenues and expenses representing the operating results from the sales and discontinuation of the Businesses.
11
ANGIODYNAMICS, INC. AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
(in thousands)
|
Aug 31, 2025
|
May 31, 2025
|
|||||||
|
(unaudited)
|
(audited)
|
|||||||
|
Assets
|
||||||||
|
Current assets:
|
||||||||
|
Cash and cash equivalents
|
$
|
38,762
|
$
|
55,893
|
||||
|
Accounts receivable, net
|
42,643
|
42,890
|
||||||
|
Inventories
|
62,255
|
62,006
|
||||||
|
Prepaid expenses and other
|
12,996
|
7,535
|
||||||
|
Total current assets
|
156,656
|
168,324
|
||||||
|
Property, plant and equipment, net
|
31,066
|
32,300
|
||||||
|
Other assets
|
9,540
|
10,404
|
||||||
|
Intangible assets, net
|
68,380
|
69,116
|
||||||
|
Total assets
|
$
|
265,642
|
$
|
280,144
|
||||
|
Liabilities and stockholders' equity
|
||||||||
|
Current liabilities:
|
||||||||
|
Accounts payable
|
$
|
31,882
|
$
|
33,291
|
||||
|
Accrued liabilities
|
27,657
|
35,518
|
||||||
|
Other current liabilities
|
8,743
|
7,388
|
||||||
|
Total current liabilities
|
68,282
|
76,197
|
||||||
|
Deferred income taxes
|
4,268
|
4,073
|
||||||
|
Other long-term liabilities
|
14,237
|
16,904
|
||||||
|
Total liabilities
|
86,787
|
97,174
|
||||||
|
Stockholders' equity
|
178,855
|
182,970
|
||||||
|
Total Liabilities and Stockholders' Equity
|
$
|
265,642
|
$
|
280,144
|
||||
12
ANGIODYNAMICS, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands)
|
Three Months Ended
|
||||||||
|
Aug 31, 2025
|
Aug 31, 2024
|
|||||||
|
(unaudited)
|
||||||||
|
Cash flows from operating activities:
|
||||||||
|
Net loss
|
$
|
(10,903
|
)
|
$
|
(12,798
|
)
|
||
|
Adjustments to reconcile net loss to net cash used in operating activities:
|
||||||||
|
Depreciation and amortization
|
6,020
|
6,785
|
||||||
|
Non-cash lease expense
|
445
|
494
|
||||||
|
Stock based compensation
|
4,470
|
3,205
|
||||||
|
Change in fair value of contingent consideration
|
—
|
76
|
||||||
|
Deferred income taxes
|
(16
|
)
|
(339
|
)
|
||||
|
Change in accounts receivable allowances
|
108
|
270
|
||||||
|
Fixed and intangible asset impairments and disposals
|
(27
|
)
|
20
|
|||||
|
Other
|
264
|
121
|
||||||
|
Changes in operating assets and liabilities:
|
||||||||
|
Accounts receivable
|
139
|
3,784
|
||||||
|
Inventories
|
(192
|
)
|
(4,053
|
)
|
||||
|
Prepaid expenses and other
|
(5,525
|
)
|
(836
|
)
|
||||
|
Accounts payable, accrued and other liabilities
|
(10,697
|
)
|
(14,982
|
)
|
||||
|
Net cash used in operating activities
|
(15,914
|
)
|
(18,253
|
)
|
||||
|
Cash flows from investing activities:
|
||||||||
|
Additions to property, plant and equipment
|
(731
|
)
|
(1,092
|
)
|
||||
|
Additions to placement and evaluation units
|
(820
|
)
|
(1,313
|
)
|
||||
|
Net cash used in investing activities
|
(1,551
|
)
|
(2,405
|
)
|
||||
|
Cash flows from financing activities:
|
||||||||
|
Principal payments on finance arrangements
|
(91
|
)
|
—
|
|||||
|
Repurchase of common stock
|
—
|
(552
|
)
|
|||||
|
Proceeds from exercise of stock options and employee stock purchase plan
|
234
|
43
|
||||||
|
Net cash provided by (used in) financing activities
|
143
|
(509
|
)
|
|||||
|
Effect of exchange rate changes on cash and cash equivalents
|
191
|
116
|
||||||
|
Decrease in cash and cash equivalents
|
(17,131
|
)
|
(21,051
|
)
|
||||
|
Cash and cash equivalents at beginning of period
|
55,893
|
76,056
|
||||||
|
Cash and cash equivalents at end of period
|
$
|
38,762
|
$
|
55,005
|
||||
13
Exhibit 99.2